Electrons + Frequency (the key idea)
- Electrons are tiny charged particles that live in atoms (especially in metals, they can move around more freely).
- Light isn’t just a wave—it also behaves like particles called photons.
- Each photon carries energy, and that energy depends on its frequency (how “fast” the wave oscillates).
The relationship is:
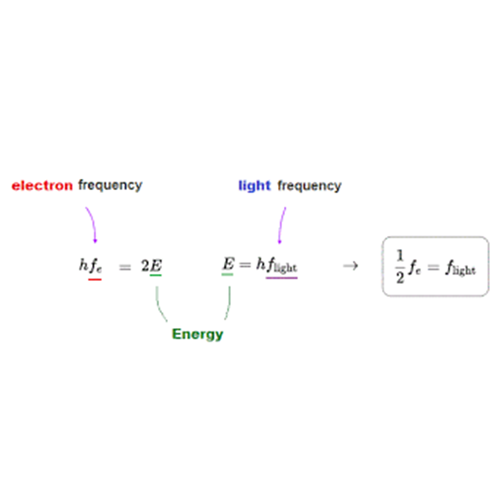
E=hfE = h f
- EE = energy of the photon
- ff = frequency of the light
- hh = Planck’s constant (just a very small number)
👉 Higher frequency = higher energy per photon
💡 The Photoelectric Effect (what actually happens)
The Photoelectric Effect is when light hits a metal and knocks electrons out of it.
Step-by-step:
- Light shines on a metal surface
- Photons hit electrons in the metal
- If the photons have enough energy, they eject electrons
- Those electrons fly off—this can even create electric current
🔑 The surprising part (this changed physics!)
Before this was discovered, scientists thought:
“Brighter light (more intensity) should knock out more electrons.”
But experiments showed something different:
✔ Frequency matters more than brightness
- If the frequency is too low → ❌ NO electrons come out, no matter how bright the light is
- If the frequency is high enough → ✅ electrons are emitted immediately
This minimum frequency is called the threshold frequency.
🧠 Why frequency matters
Think of it like this:
- Each electron needs a minimum energy to escape the metal (called the work function)
- A photon either has enough energy… or it doesn’t
👉 It’s not about “building up energy” over time
👉 It’s a one-hit event: one photon hits one electron
🔥 What happens when frequency increases?
- Higher frequency → higher photon energy
- Electrons come out with more speed (kinetic energy)
⚡ The key equation for the effect
Ephoton=work function+kinetic energy of electronE_{text{photon}} = text{work function} + text{kinetic energy of electron}
Or:
hf=ϕ+KEhf = phi + KE
👨🔬 Why this matters
The photoelectric effect was explained by Albert Einstein, and it was a big deal because it showed:
- Light behaves like particles, not just waves
- Energy comes in discrete packets (quanta)
This helped launch quantum physics.
🧩 Simple analogy
Imagine electrons are balls in a pit:
- Low-frequency light = weak toss → can’t get the ball out
- High-frequency light = strong toss → ball escapes instantly
Brightness just means:
- More balls being thrown, not stronger throws
✔ Quick summary
- Light energy depends on frequency, not brightness
- Electrons are only ejected if frequency is high enough
- Each photon interacts with one electron
- This is proof that light has particle behavior